Agricultural and Biological Research
RNI # 24/103/2012-R1
Research - (2022) Volume 38, Issue 5
Population increase and rapid urbanization are two major factors for scarcity of clean water in 21st century societies across the world. Modern sewage water treatment plants and filtration systems require a lot of economic and land resources which many “lesser developed countries” do not have, leading to large amounts of raw sewage water being released into the environment and a lack of clean water for millions. To fulfill such water demand, the use of aquatic plants, also known as macrophytes, have been used in a constructed wetland which allows for the filtration of contaminated water using natural processes involving wetland vegetation, soils, and their associated microbial assemblages. In the present study, Typha (cattail) and Pontederia crassipes (water hyacinth) have been tested in a simulated constructed wetland to study their effect on pollutants removal efficiency (nitrates, ammonium, pH, and dissolved oxygen). Results show that both the Typha and Pontederia crassipes were effective in filtering out the aforementioned contaminants through the process of phytoaccumulation. However, there was no clear correlation between dissolved oxygen levels and time in contact with the macrophyte roots possibly due to environmental factors. A better quantitative understanding of the phytoaccumulation process, such as the limits and plateau points of contaminant uptake is needed to optimize the usage and application of Typha and Pontederia crassipes in practical constructed wetlands.
Aquatic plants; Phytoaccumulation; Typha; Pontederia crassipes
All living things – plants, animals, and human beings require clean water. Good water quality is essential to human health, the economy, and our ecosystem. However, as populations grow rapidly and natural environments degrade, ensuring sufficient and safe water supplies for everyone has become increasingly challenging. It is estimated that each person uses around 100 gallons of water per day (United States Geological Survey, 2019). The used water turns into sewage water and then flows down pipes and drains and into sewers that are located underneath roads. The sewage is then carried to waste treatment centers, where it is screened for bacteria, chemicals, floating objects, and other major water pollutants. Building these pipes and sewage water treatment plants for a water filtration system requires a lot of money and resources, especially energy resources, which many developing countries in Southeast Asia and Africa lack. As a result, large quantities of raw wastewater are released directly into rivers and oceans in these areas. According to the United Nations Sustainable Development Goal 6 Water Report in 2021, 44% of household wastewater is not safely treated globally, and over 80% of wastewater is released into the environment without adequate treatment. In addition, the World Health Organization estimates that every year more than 3.4 million people die as a result of water related diseases, making it the leading cause of disease and death around the world (World Health Organization, 2009). In order to solve this problem, we must reduce our pollution output and improve the way we manage wastewater so it can be safely released into rivers and water sources without damaging the environment and our communities.In this research, we will be determining the effectiveness of a constructed wetland in the filtration of sewage water. In order to monitor clean levels of water, the following variables will be tested: Dissolved oxygen (DO), pH, Ammonia (NH3) and Nitrate (NO3). A constructed wetland is a natural process involving wetland vegetation, soils and their associated microbial assemblages to improve water quality. It is a lot cheaper and requires less energy than standard water filtration and sewage systems that many developed countries use. It is also more environmentally friendly as it only requires natural processes to filter contaminated water. The concept of the constructed wetland was developed in the 1960s by Käthe Seidel in Germany. Before that, between 1952 and 1956, Seidel carried out numerous experiments on the use of wetland plants for the treatment of various types of wastewater, including phenol, dairy, or livestock wastewater [1]. The ability of wetlands to transform and store organic matter and nutrients has resulted in the widespread use of wetlands for wastewater treatment worldwide. Wetland plants are an important component of wetlands, and the plants have several roles in relation to the wastewater treatment processes. The filtration of water contaminants in a constructed wetland relies on the plant’s root systems and plant tissues, particularly the processes of phytoremediation and phytoaccumulation where the plants uptake contaminants and particles as they use that as their nutrients to grow and perform photosynthesis [2]. The plant tissues are responsible for filtering out large pieces of debris, providing a surface area for attached biofilms, an excretion of photosynthetic oxygen, which leads to increased aerobic degradation and up taking nutrients from the water. Within the plant tissues, there are dense amounts of photosynthetic algae, bacteria, and protozoa. Likewise, the roots and rhizomes that are buried in the wetland soil provide a substrate for the attached growth of microorganisms (Hofmann, 1986). These biofilms along with including dead macrophyte tissues are responsible for the majority of the microbial processing that occurs in wetlands. Later researchers have conducted experiments testing the effectiveness of constructed wetlands in various parts of society, such as putting sewage water from agricultural, industrial, petrochemical, and power plant industries into a constructed wetland to determine if water quality improved after the process. In addition, researchers have also tested various aquatic plants, also known as macrophytes, such as reed (Phragmites australis), water spinach (Ipomoea aquatica), bulrush (Scirpus), Timothy grass (Phleum pratense) and bamboo (Phyllostachys heteroclada) to determine their root system’s effectiveness in filtering contaminated water.
In most published experiments related to constructed wetlands and filtration, scientists conduct the experiment at the locations near where the wastewater is being discharged, such as in lakes and river basins near the site of wastewater discharge. This condition will make wastewater constantly flow through the filtration site along with natural factors such as the sun, wind, and other animals which will impact the water quality being filtered by the macrophytes in those filtration sites. In this experiment, there will be a natural wastewater sample taken from the Drunken Moon Lake located in the National Taiwan University. This means the macrophytes used in this experiment will be filtering non-flowing water rather than flowing water, which will make the amount of Dissolved oxygen and contaminants in the water less compared to flowing water, which will potentially have an impact on the number of contaminants that the macrophytes will be able to filter out and will improve on the findings of other researchers in this field. Moreover, in previous experiments done by researchers in this field, the Typha that was used has leaves on the Typha, which will allow it to perform photosynthesis and have a stronger and more developed root system and plant tissues.
However, the Typha used in this experiment does not have leaves, meaning the filtration by the root system and the plant tissues of the Typha will not be as strong compared to the Typha with leaves, which will be determined in this experiment. Furthermore, the Pontederia crassipes are considered as a novelty in constructed wetland experiments, which will make the findings of this experiment critical to the use and effectiveness of Pontederia crassipes in a constructed wetland (Figures 1 and 2).

Figure 1: Pontederia crassipes habitat.

Figure 2: Typha habitat placed in a water tank.
Experimental setup
In this experiment, two macrophytes will be used: cattail (Typha) and water hyacinth (Pontederia crassipes). The macrophytes will be isolated in their own individual plant systems in order to determine the effectiveness of each of their root systems in filtering contaminants from contaminated wastewater. The water quality is taken by using Vernier sensor probes.
Construction of the macrophyte habitat
20 Typha and 10 Pontederia crassipes were obtained from a local plant shop. The Typha was placed in a 1.5 L water tank filled with 1.2 L of water as their habitat to grow and live in. A 20 cm × 30 cm styrofoam lid with 20 holes of a diameter of 1.5 cm was made in order to put the Typha in the 1.5 L water tank while maintaining upright posture and not collapsing, ensuring the roots are sufficiently hydrated and are able to grow. The Pontederia crassipes was placed individually in a 5.0 L tank filled with enough water to cover the roots while maintaining the macrophyte in an upright position. Both containers received lighting from a LED lightbulb during the day for 8 hours to simulate sunlight so the macrophytes can perform photosynthesis and grow.
Obtaining contaminated water
The contaminated water is taken from a natural wastewater sample taken from the Drunken Moon Lake located in the National Taiwan University, which contains the contaminants, Dissolved Oxygen (DO), pH, Ammonia (NH3) and Nitrate (NO3). 5 L of the contaminated water is collected to be used in the experiment for each trial.
Conducting experiment
One Typha and one Pontederia crassipes will be taken from the 1.5 L tank and placed in a separate 4 L beaker filled with 3 L of sample contaminated water from the Drunken Moon Lake for each macrophyte. A big Pontederia crassipes represents Pontederia crassipes with a root diameter (measured from the end of the stem) larger than 10 cm, while a small Pontederia crassipes represents Pontederia crassipes with a root diameter of less than 10 cm. After the contaminated water has been poured into the beaker, a water quality sample will be taken from each of the respective macrophyte water beakers by inserting a Vernier Dissolved Oxygen Probe, Vernier pH Sensor, Vernier Ammonium Ion-Selective Electrode and a Vernier Nitrate Ion-Selective Electrode into the macrophyte container to determine the amounts of the listed amounts within the water sample. All vernier probes were calibrated according to the sample solutions provided in the Vernier user manual. The testing probes will then be placed inside the macrophyte’s water beaker to take a measurement. The water samples will be taken in intervals of 1 hour until the 4 hours mark and then the next measurement will be taken after 24 hours. Then, the remaining measurements will be taken in intervals of 24 hours, meaning the measurement after the one taken after 24 hours is after 48 hours after the contaminated water has been poured into the beaker. The experiment will last for 3 days (72 hours). The results of the water sample test will be recorded and analyzed to determine the amount of each contaminant that has been filtered by each of the macrophytes.
Across the board, all macrophytes were effective in filtering out contaminants and purifying the contaminated water solution.
Nitrates
As seen in Figure 3, it is evident that the nitrate concentrations decreased as time increased after in contact with each respective macrophyte. According to the United States Environmental Protection Administration (USEPA), safe levels of nitrates for communal wastewater cannot exceed 10 mg/L. The initial concentration of nitrates was 0.2 mg/L, which is quite low compared to normal wastewater samples. However, this wastewater sample was collected from the Drunken Moon Lake, which has already been filtered by the Taiwanese water filtration system. Nevertheless, after an hour, all of the nitrate concentrations dropped dramatically to around 0.1-0.15 mg/L. After 2 hours, most of the macrophytes were able to absorb and uptake all of the nitrates to make the concentration of nitrates in the water sample 0 mg/L, which shows that each different macrophyte was effective in filtering out the nitrates. These findings are supported by SEM images taken before and after the filtration process.
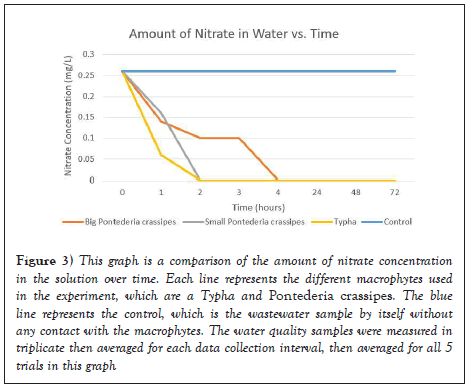
Figure 3: This graph is a comparison of the amount of nitrate concentration in the solution over time. Each line represents the different macrophytes used in the experiment, which are a Typha and Pontederia crassipes. The blue line represents the control, which is the wastewater sample by itself without any contact with the macrophytes. The water quality samples were measured in triplicate then averaged for each data collection interval, then averaged for all 5 trials in this graph
As seen in Figure 4, there are some white many crystals covering the roots of the plant. According to the elemental analysis tool, the white crystals were composed mostly of carbon and oxygen, which is expected in an aquatic plant as plants as plants are autotrophs. This means they produce their own food using the process of photosynthesis to transform water, sunlight, and carbon dioxide into oxygen. Some of the carbon is used for plant growth, and some of it is used in respiration, where the plant breaks down sugars to get energy [3]. However, as seen in Figure 5, it is evident that nitrates were absorbed by the macrophytes as the elemental analysis chart shows a higher potassium concentration in the Pontederia crassipes root after being in a KNO3 solution for 72 hours. The potassium cation remains in the root while the nitrate anion was absorbed. This is a process a process known as phytoaccumulation where the plants uptake contaminants and particles as they use that as their nutrients to grow and perform photosynthesis. Phytoaccumulation is when the roots of the plant uptake contaminants along with other nutrients and water. The contaminant mass is not destroyed but ends up in the plant shoots and leaves that can be harvested for disposal and growth. The results of this experiment supports that aquatic plants are effective in performing phytoaccumulation and uptaking nitrates from water solutions, which is crucial in water filtration as consuming too much nitrate can affect how blood carries oxygen and can cause methemoglobinemia, especially for babies under six months old. Recent research indicates more health hazards associated with nitrate exposure and other health effects such as increased heart rate, nausea, and abdominal cramps. Some studies also suggest an increased risk of cancer, especially gastric cancer associated with dietary nitrate exposure, but there is not yet a scientific consensus on the previously mentioned claim.

Figure 4: This is an image of a control Pontederia crassipes root rested in tap water taken from a Scanning Electron Microscope (SEM). The root was not in contact with any contaminants, and some visible crystals are seen which are likely particles from the tap water. The chart on the right is an elemental analysis chart from the SEM image of the control Pontederia crassipes. The x axis represents the Energy in keV, and the y axis represents the counts per second. The elemental analysis chart shows that the section marked in blue, which includes the white crystals, are mostly made up of carbon and oxygen which is expected in a macrophyte.
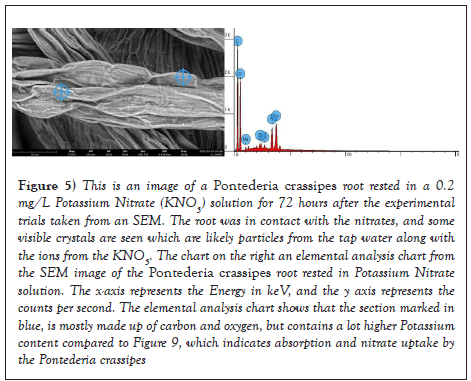
Figure 5: This is an image of a Pontederia crassipes root rested in a 0.2 mg/L Potassium Nitrate (KNO3) solution for 72 hours after the experimental trials taken from an SEM. The root was in contact with the nitrates, and some visible crystals are seen which are likely particles from the tap water along with the ions from the KNO3. The chart on the right an elemental analysis chart from the SEM image of the Pontederia crassipes root rested in Potassium Nitrate solution. The x-axis represents the Energy in keV, and the y axis represents the counts per second. The elemental analysis chart shows that the section marked in blue, is mostly made up of carbon and oxygen, but contains a lot higher Potassium content compared to Figure 9, which indicates absorption and nitrate uptake by the Pontederia crassipes.
Ammonium
As seen in Figure 6, ammonium concentrations decreased significantly after a macrophyte was placed in the water sample. According to the USEPA and WHO, there is no established maximum contaminant level in communal and drinking water. However, the National Academy of Science recommends, and many European nations have adopted, a drinking water standard of 0.5 mg/L. Initially, the ammonium concentration was 2.0 mg/L, but after 1-2 hour in contact with each of the respective macrophytes, the ammonium concentration decreased to around 1.0-1.5 mg/L. As time increased, the ammonium was absorbed by the macrophyte roots through the process of phytoaccumulation, and eventually decreased to 0 mg/L after 24 hours.
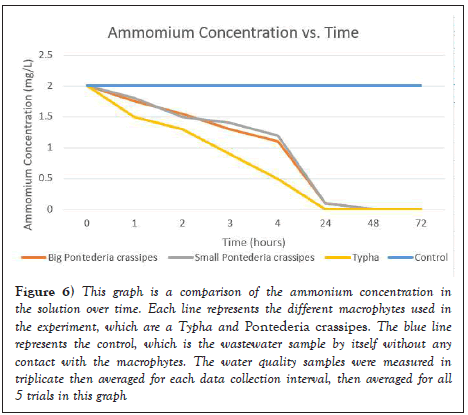
Figure 6: This graph is a comparison of the ammonium concentration in the solution over time. Each line represents the different macrophytes used in the experiment, which are a Typha and Pontederia crassipes. The blue line represents the control, which is the wastewater sample by itself without any contact with the macrophytes. The water quality samples were measured in triplicate then averaged for each data collection interval, then averaged for all 5 trials in this graph.
This is crucial as ammonia is toxic and detrimental to most fish and other aquatic organisms at concentrations above 1 mg/L in water. Similarly, long-term ingestion of water containing more than 1 mg/L may damage the internal organ systems and even cause severe burns and scarring to humans (Oregon Department of Human Services, 2000).
pH
As seen in Figure 7, pH levels decreased after in contact with each macrophytes. According to the USEPA, safe levels of pH for communal wastewater should be between 6.0-8.5. The original concentration of the wastewater sample was around 10.2. However after 1 hour in contact with the macrophytes, the pH levels decreased significantly to around 8-9, and began to plateau at around 7.6 as time went on which is within the safe levels of pH in water.
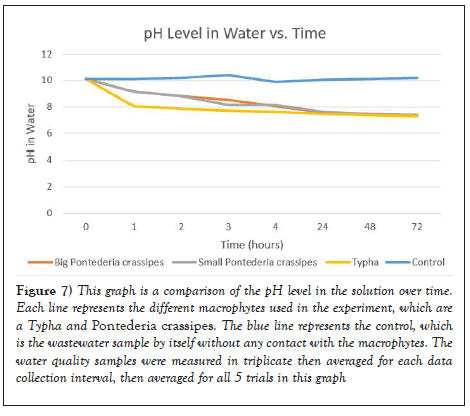
Figure 7: This graph is a comparison of the pH level in the solution over time. Each line represents the different macrophytes used in the experiment, which are a Typha and Pontederia crassipes. The blue line represents the control, which is the wastewater sample by itself without any contact with the macrophytes. The water quality samples were measured in triplicate then averaged for each data collection interval, then averaged for all 5 trials in this graph.
This is because plants can metabolize several other chemicals and change the pH. In the photosynthesis process, plants absorb carbon dioxide and produce oxygen. Carbon dioxide will lower the pH of water, as when CO2 is dissolved in water, a part of it reacts with water to form carbonic acid (H2CO3). The hydrogen ions present in carbonic acid will make water acidic, meaning that the pH will be lowered. Thus, when aquatic plants absorb carbon dioxide and remove it from water, the pH will rise slightly as there is less hydrogen ions present in the water [4]. On the other hand, plants also absorb ammonia and other nitrogen compounds. These compounds cause biological reactions that produce free hydrogen ions, which lower the pH as the formula for calculating pH is -log [H⁺]. This has a much larger effect on pH compared to the aforementioned effect of plants absorbing carbon dioxide in water [5]. Thus by absorbing these nutrients and contaminants, macrophytes could possibly prevent the pH from increasing too high (Figure 8).
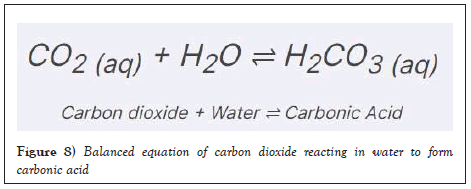
Figure 8: Balanced equation of carbon dioxide reacting in water to form carbonic acid.
Dissolved oxygen (DO)
According to the Taiwan EPA, safe levels of DO for communal wastewater should be above 6.50 mg/L. As seen in Figure 9, the initial concentration of Dissolved oxygen in the water sample was 8.03 mg/L. After 1 hour in contact with each of the respective macrophytes, the DO levels increased from 8.03 mg/L to 8.15 mg/L in the big Pontederia crassipes and 9.21 mg/L in the small Pontederia crassipes. However, the DO amount in the Typha decreased to 6.63 mg/L, significantly less compared to the Pontederia crassipes. The control in the experiment as represented by the blue line indicates that there is no clear correlation between DO levels and time when in contact with a macrophyte. This is likely due to environmental factors as the amount of oxygen that can be dissolved in water depends on water temperature, the amount of dissolved salts present in the water which is salinity, and the atmospheric pressure [6]. Nevertheless, these variables accurately reflect the practical usage of macrophytes in a constructed wetland as the environmental factors may vary daily and thus lead to mixed results in DO concentration in water. In addition, different macrophytes can have different impacts on DO concentration. Oxygen release rates from roots depend on the internal oxygen concentration, the oxygen demand of the surrounding medium and the permeability of the root-walls [7]. Macrophytes release oxygen from their roots as rates of oxygen leakage are generally highest in the sub-apical region of roots and decrease with distance from the root-apex [8]. In addition, research shows that aquatic plants with an internal convective throughflow ventilation system have higher internal oxygen concentrations in the roots compared to aquatic plants that rely on the diffusive transfer of oxygen [9]. Thus, aquatic plants with a convective through flow mechanism, such as the Typha, have the potential to release more oxygen from their roots compared to species without convective through flow which will impact the DO concentration in water as seen in the results of the experiment (Figures 9 and 10 ) [10,11].
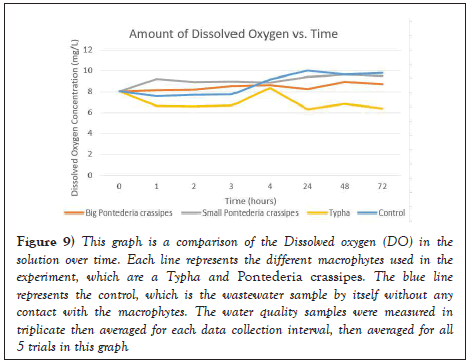
Figure 9: This graph is a comparison of the Dissolved oxygen (DO) in the solution over time. Each line represents the different macrophytes used in the experiment, which are a Typha and Pontederia crassipes. The blue line represents the control, which is the wastewater sample by itself without any contact with the macrophytes. The water quality samples were measured in triplicate then averaged for each data collection interval, then averaged for all 5 trials in this graph.

Figure 10: The image on the left is the original wastewater sample without any filtration by macrophytes. The image on the right is the water sample after filtration by a Typha for 72 hours.
In this study, we attempt to find and improve on the effectiveness of the Typha and Pontederia crassipes in filtering contaminants from wastewater. As evident from all graphs from the experiments, there have been improvements in water quality in terms of nitrates, ammonium, and pH levels after the contaminated water solution was put into the root systems of each respective. However, there is no clear correlation between DO levels and time when filtered by a macrophyte.
These conclusions are supported by figure 12, which shows that the water quality improved significantly as the filtered water was less opaque and clear, which indicates that contaminants have been removed. The Typha was notable in being the most effective as it filtered contaminants at a very high rate. This is likely because Typha roots have a larger diameter compared to Pontederia crassipes roots in general, which allows it to uptake contaminants more efficiently. It may also be because the Typha have stronger capillarity action compared to other macrophytes, meaning that the plant cells can pull water and contaminants along with it stronger and faster in the phytoaccumulation process, but more research is needed to verify that conclusion.
In the future, potential avenues for further research are to better understand the cellular mechanism on how macrophytes can trap contaminants by using microscopic images of the phytoaccumulation process and to figure out a specific plateau point for contaminant absorption for various macrophytes. This information is crucial to workers in the agricultural industry and the government as they can use this information to apply these aquatic plants to a practical setting. Moreover, developing further research on the effectiveness of various macrophytes in filtering contaminated water is crucial as different areas of the world have different climates and allow the growth of different plants and macrophytes. By having a more developed knowledge on the potential macrophytes to use in a constructed wetland, many countries can begin using this environmentally friendly and highly effective method in providing safe and clean water for their citizens.
Special thanks to Mr. Jude Clapper for demonstrating laboratory procedures and guidance in this research. Special thanks to Mr. Sean Tsao for helping with orders of materials needed for this research. This scientific work makes use of the Taipei American School Science Laboratory, operated by the Department of Scientific Research.
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript. These authors contributed equally.
[Crossref]
[Crossref] [Google Scholar] [Pubmed]
[Crossref] [Google Scholar] [Pubmed]
Citation: Moreira H, Ross G. Investigation of the effectiveness of aquatic plants in filtering contaminated water. AGBIR.2022; 38(5): 366-370.
Received: 18-Jul-2022, Manuscript No. AGBIR-22-69499; , Pre QC No. AGBIR-22-69499 (PQ); Editor assigned: 21-Jul-2022, Pre QC No. AGBIR-22-69499 (PQ); Reviewed: 04-Aug-2022, QC No. AGBIR-22-69499; Revised: 11-Aug-2022, Manuscript No. AGBIR-22-69499 (R); Published: 18-Aug-2022, DOI: 10.35248/0970-1907.22.38.366-370
Copyright: This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http:// creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
